Abstract/References
Successful management of unstable angina in a ravulizumab-treated patient with paroxysmal nocturnal hemoglobinuria
Hiroshi Takahashi, Hirotaka Mori, Masahiko Fukatsu, Takahiro Sano, Kayo Harada, Masayoshi Oikawa, Yasuchika Takeishi, Satoshi Kimura, Hiroshi Ohkawara, Tsutomu Shichishima, Takayuki Ikezoe
Author information
- Hiroshi Takahashi
Department of Hematology, Fukushima Medical University - Hirotaka Mori
Department of Hematology, Fukushima Medical University - Masahiko Fukatsu
Department of Hematology, Fukushima Medical University - Takahiro Sano
Department of Hematology, Fukushima Medical University - Kayo Harada
Department of Hematology, Fukushima Medical University - Masayoshi Oikawa
Department of Cardiology, Fukushima Medical University - Yasuchika Takeishi
Department of Cardiology, Fukushima Medical University - Satoshi Kimura
Department of Hematology, Fukushima Medical University - Hiroshi Ohkawara
Department of Hematology, Fukushima Medical University - Tsutomu Shichishima
Department of Hematology, Fukushima Medical University - Takayuki Ikezoe
Department of Hematology, Fukushima Medical University
Abstract
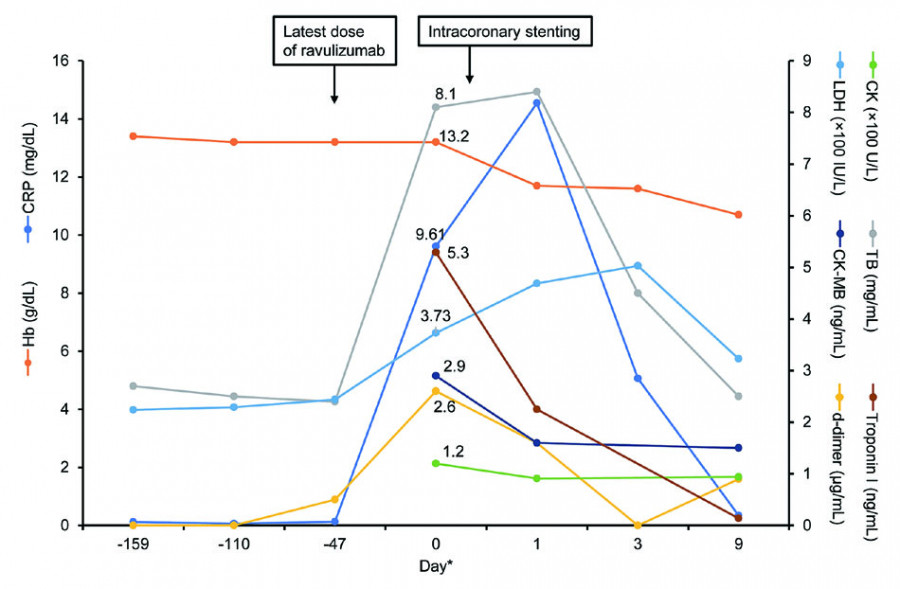
Ravulizumab is an anti-C5 antibody approved for treating paroxysmal nocturnal hemoglobinuria (PNH). In August 2019, a 77-year-old Japanese man with PNH, who had been on ravulizumab treatment for 2 years, was hospitalized for chest discomfort and malaise. Electrocardiography identified a right bundle block, and elevated serum troponin I and d-dimer suggested ischemic heart disease. Cardiac catheterization revealed severe stenosis in the left anterior descending coronary artery, and intracoronary stenting relieved his chest discomfort. The final diagnosis was unstable angina unrelated to ravulizumab, and the patient’s ravulizumab treatment was uninterrupted with no significant complications of PNH. This case report highlights the importance of continuing complement inhibition therapy during acute coronary events.
References
- 2.Hill A, DeZern AE, Kinoshita T, Brodsky RA. Paroxysmal nocturnal haemoglobinuria. Nat Rev Dis Primers, 3:17028, 2017.
- 9.Harder MJ, Kuhn N, Schrezenmeier H, et al. Incomplete inhibition by eculizumab:mechanistic evidence for residual C5 activity during strong complement activation. Blood, 129:970-980, 2017.
- 11.Connell NT. Ravulizumab:a complementary option for PNH. Blood, 133:503-504, 2019.
Figures (1)