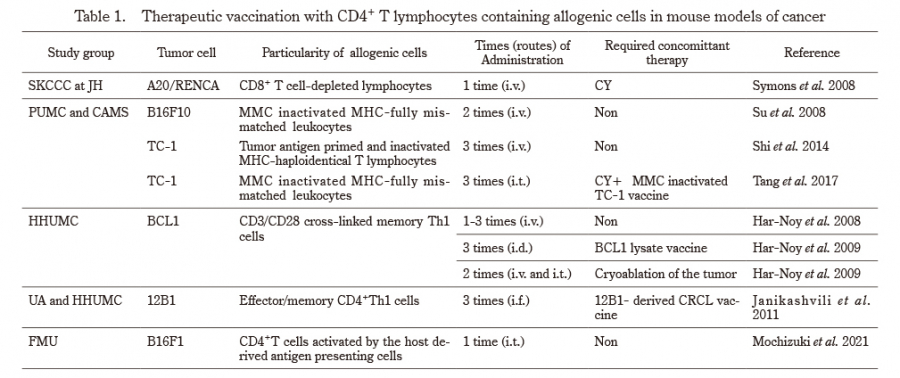
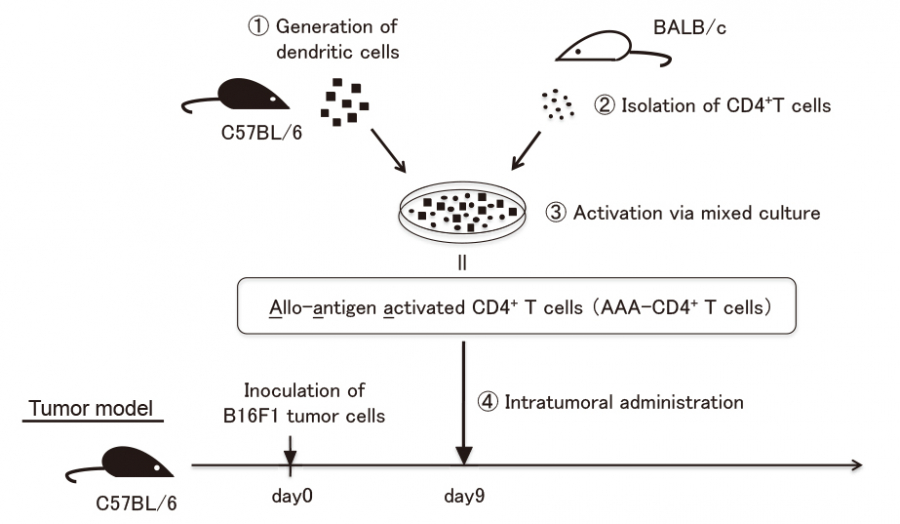
Advanced cancer has a high mortality rate and is one of the most difficult diseases to cure. Immunotherapies developed over the past decade have been among the most promising approaches for the treatment of patients with advanced cancer1). Recent findings suggest that cancer cells shield themselves by expressing immune checkpoint molecules, such as PD-L1, which bind to the PD-1 receptors expressed on activated T cells, resulting in the loss of their capacity to attack cancers2,3). The blockade of these immune checkpoint molecules using either anti-PD-1 antibody (Ab) or anti-PD-L1 Ab can reinvigorate the host endogenous antitumor immunity4,5). Based on their clinical efficacy, these immune checkpoint inhibitors have been approved for many cancers, including melanoma, non-small cell lung cancer, and renal cell carcinoma3-8). However, the overall objective response rate of these therapies based on immune checkpoint blockade rarely exceeds 40% in various cancers3,9,10). On the other hand, blocking physiological immunosuppressive pathways such as PD-1/PD-L1 interactions often causes serious autoimmune-like adverse reactions11,12). Therefore, novel strategies are required to improve the efficacy of immunotherapy for advanced cancers.
Adoptive cell therapy is another innovative approach for treating advanced malignancies. Engineering human T cells with a chimeric antigen receptor (CAR) specific for the pan-B-cell CD19 antigen (CD19 CAR-T cells) has been associated with high response rates in patients with relapsed or refractory B-cell malignancies13). Many other types of CAR-T cell therapies have been developed to target various antigens presented on the surface of both hematological malignant cells and non-hematological solid tumors14-16). However, CAR-T cell therapies in solid tumors are not as effective as those in leukemia because of the paucity of preferable target antigens, as well as their inability to infiltrate and survive in the tumor microenvironment17,18). Furthermore, one of the challenges of these target-specific therapies is relapse due to emerging clones that have lost their target antigens13,19). Indeed, other target-specific immune therapies, such as bispecific antibodies and several monoclonal antibodies, have the same problem of relapse due to the loss of target antigens20,21). In this context, Kuhn et al. demonstrated that CD40 ligand-modified CAR-T cells not only elicited antitumor activity but also circumvented tumor immune escape by inducing endogenous antitumor immunity22,23). Li et al. also demonstrated that CAR-T cells secreting IL-36γ activate endogenous antigen-presenting cells (APCs) and T cells, which improves CAR-T cell-mediated antitumor responses24). These results suggest that endogenous antitumor immunity in concert with CAR-T cells may play a pivotal role in overcoming escaped clones and eliciting potent antitumor effects.