Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, non-neoplastic, acquired hematopoietic stem cell disorder1). Eculizumab, a human monoclonal antibody that blocks complement protein C5, is now considered the standard of care for treating PNH2,3). Although it is highly effective, approximately 11%-27% of patients who receive approved dosages of eculizumab for PNH experience potentially life-threatening breakthrough hemolysis (BTH)4-6). BTH is defined as at least one new or worsening symptom or sign of intravascular hemolysis (fatigue, hemoglobinuria, abdominal pain, dyspnea, anemia [hemoglobin <10 g/dL], major adverse vascular event [including thrombosis], dysphagia, or erectile dysfunction) in the presence of elevated lactate dehydrogenase (LDH) ≥2× the upper limit of normal (ULN) after prior reduction of LDH to <1.5×ULN on treatment7,8). Causes of BTH include suboptimal inhibition of C5 and/or complement-amplifying conditions that arise due to infection, surgery, or pregnancy9,10).
Ravulizumab is a newly developed, human anti-C5 antibody derived from eculizumab, with an 8-week dosing interval11). Ravulizumab demonstrated noninferiority to eculizumab for the endpoints, including LDH normalization and incidence of BTH, in two international phase 3 trials (including Japan); one in complement inhibitor-naïve patients (study 301 [NCT02946463])7) and the other in eculizumab-experienced patients (study 302 [NCT03056040])8). In study 301, five non-Japanese patients experienced BTH while receiving ravulizumab despite complete blockade of free C5 (free C5 <0.5 µg/mL) and 13 experienced BTH during treatment with eculizumab7,12). By comparison, in study 302, none of the patients who were stable on eculizumab and then switched to ravulizumab developed BTH while receiving ravulizumab and none experienced thromboembolic events;but five non-Japanese patients who received eculizumab experienced seven BTH events8,12).
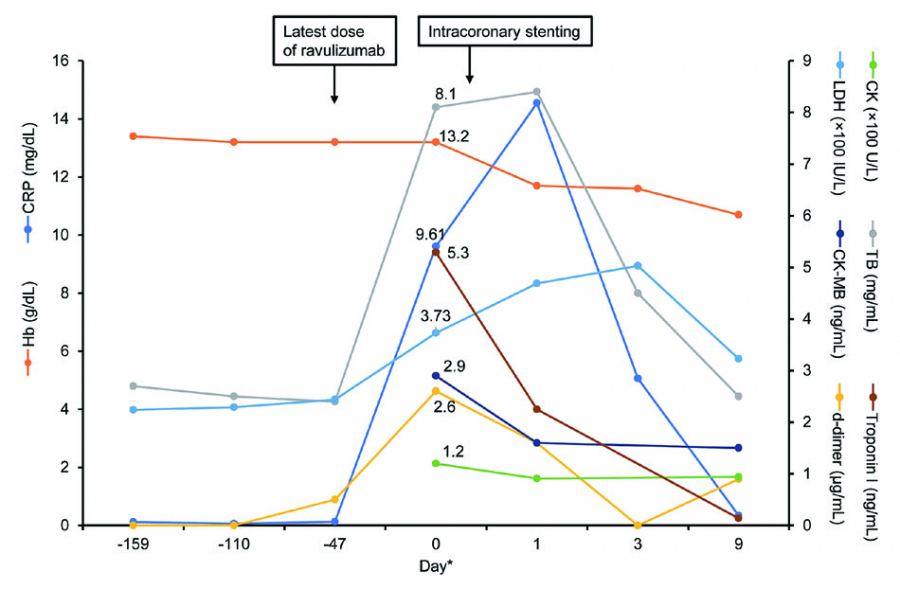
Situations like infection, pregnancy, or surgery could increase complement activation placing patients on eculizumab or ravulizumab at risk of BTH. Here, we describe our experience of a 77-year-old male Japanese patient with PNH who was successfully continued on ravulizumab during an acute event of unstable angina (non-ST-elevation myocardial infarction).