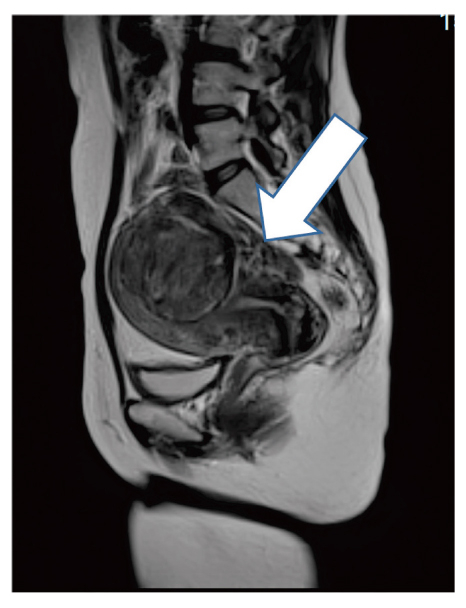
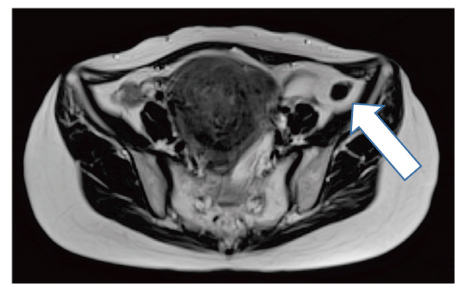
Uterine smooth muscle tumors with unusual growth patterns include three primary neoplasms:intravenous leiomyomatosis (IVL), benign metastasizing leiomyoma (BML), and leiomyomatosis peritonealis disseminata (LPD)7). IVL is defined as the pathological presence of smooth muscle cells growing in venous or lymphatic vessel spaces without invading them7). The plausible etiology of IVL comprises uterine leiomyomas entering the venous system or directly originating from the walls of the uterine veins8,9) BML is characterized by presence of multiple smooth muscle nodules frequently located in the lung, lymph nodes, or abdomen. Although the etiology of BML is unknown, the most plausible theory proposes that BML might occur secondary to iatrogenic implantation due to morcellation of leiomyoma or proliferation of benign smooth-muscle tissue, embolized by an IVL10). Mostly, BML is asymptomatic or diagnosed when acute complication such as chest pain, dyspnea, and cough due to development of pulmonary lesions7). LPD is a rare benign disease that occurs in women of reproductive age. In most patients, LPD is asymptomatic and discovered during surgery performed for other conditions7). The lesion is characterized by the presence of multiple smooth muscle nodules on the abdominal, pelvic peritoneal surfaces and sometimes confused with peritoneal carcinomatosis or tuberculous peritonitis7).
PM was first reported by Willson and Peale in 195211). Since then, approximately 130 cases in the English-language literature have been reported4). Apart from the above three unusual smooth muscle cell growing patterns, secondary or iatrogenic PM is the most common form of PM and seen as a complication of previous myomectomy, particularly following laparoscopy using a morcellator. Previous reports on parasitic fibroids are sparse, and most reported cases have been linked to previous laparoscopic surgery4,11). Unlike previous reports, the present case had no history of previous surgery. Therefore, we considered that PM without a history of previous uterine surgery could be identical with LPD.
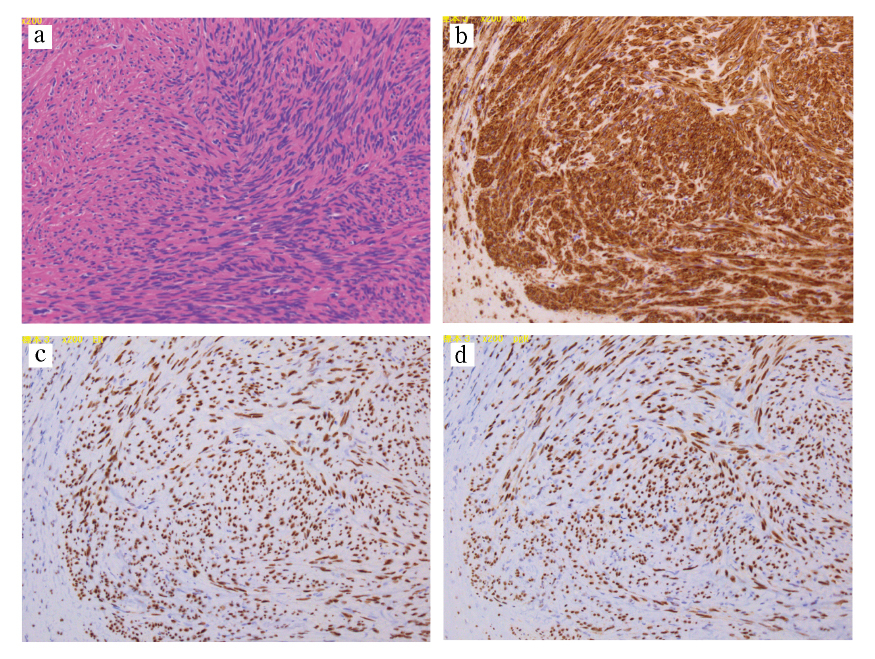
Although there have been a few theories, the exact pathophysiology of PM, other than iatrogenic factors, remains unclear. Hormonal stimulation is thought to be a key contributor in the pathophysiology of PM. Tavassoli et al also postulate that PM occurs due to an unusual predisposition of subperitoneal mesenchymal stem cells12). These processes are usually stimulated by estrogen12). This hypothesis is supported by cases of PM after the use of oral contraception13), hormone replacement therapy14), tamoxifen15), and ovarian stimulation treatment16). However, our patient received anti-hormonal therapy prior to undergoing TLH. As in our case, other reports describe the occurrence of PM without hormonal exposure, including a report of seven postmenopausal patients with PM, none of whom received hormone replacement therapy17). Based on these contradictory conclusions, the etiology of non-iatrogenic PM remains unclear. Although PM is usually a benign condition, nine cases of malignant transformation have been reported17). Among them, seven cases occurred in women of reproductive age17). Histopathological lesions, such as cytologic atypia, nuclear polymorphism, hyperchromasia, tumor cell necrosis, and increased mitotic Figures, are signs of malignant transformation17).
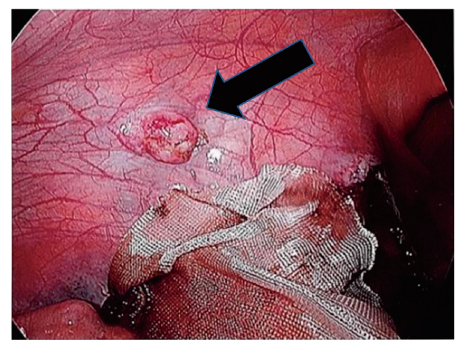
We perform laparoscopic surgery for an enlarged uterus in many cases, which allows for a detailed examination of various pelvic and abdominal conditions. Consequently, we found that PM could not be identified prior to performing surgery. We suggest that laparoscopic surgery enables the identification of small tumors growing in the peritoneal area.
In conclusion, we successfully managed uterine myoma and PM using laparoscopic surgery. Regardless of a history of previous myomectomy, PM should be considered in women with uterine myoma. This report also warrants further discussion and research into the biological characteristics of uterine myomas.