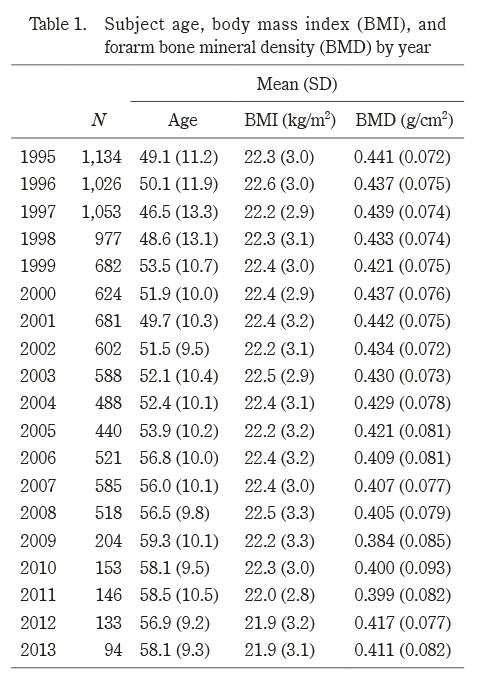
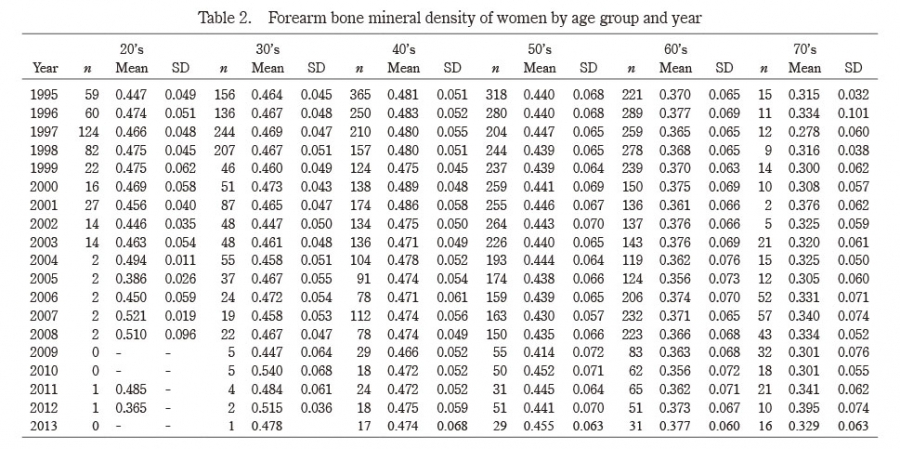
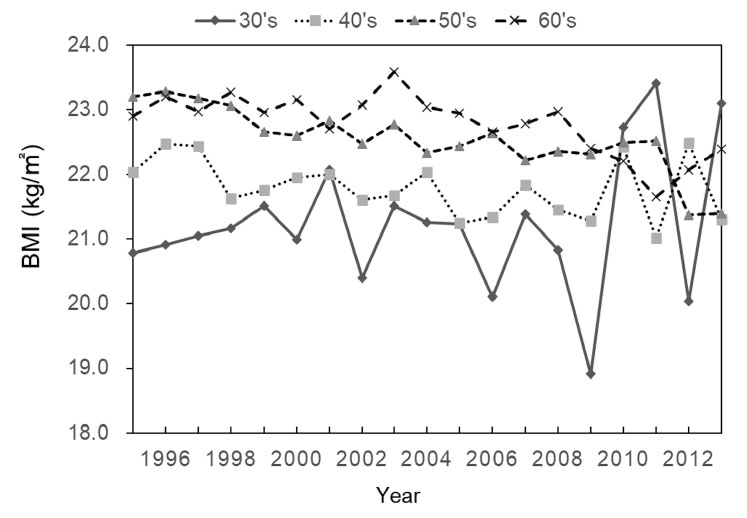
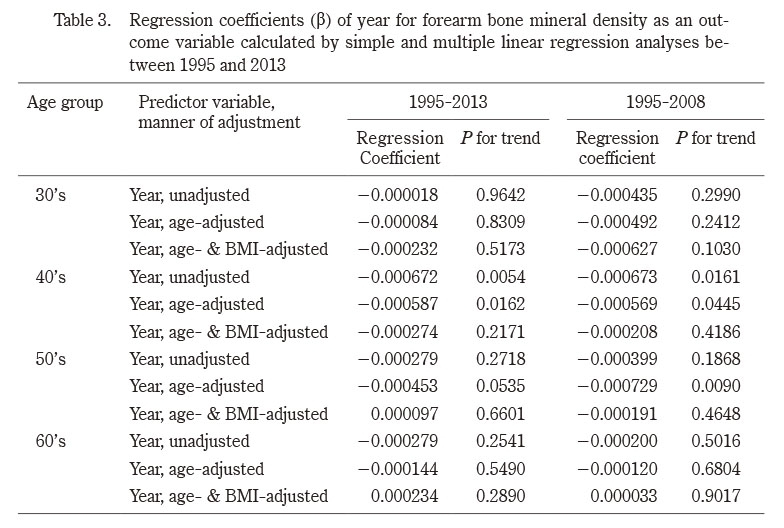
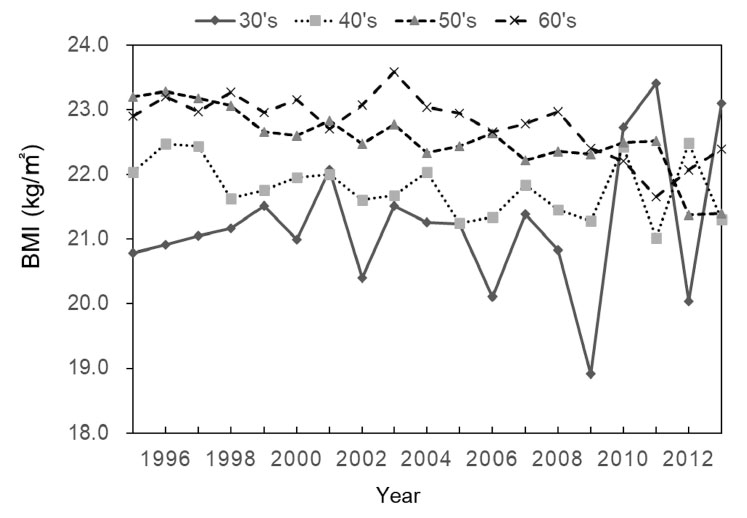
The present study investigated secular changes in forearm BMD and BMI of adult Japanese women from 1995 to 2013, with the following findings: 1) age-adjusted BMD of subjects in their 40’s and 50’s decreased, 2) their age- and BMI-adjusted BMD did not decrease, and 3) BMI of subjects in their 40’s, 50’s, and 60’s decreased.
Only a few studies have reported on secular changes in BMD. Xu and Wu7) studied hip BMD in US subjects (age ≥30 years [mean age, 53 years]) who participated in the NHANES from 2005 to 2014, and found that their BMD decreased during that period, particularly between 2009 and 2014. The authors suggested that this decrease in BMD corresponded to an increased trend of hip fracture in women aged ≥65 years between 2012 and 20156), and that BMD is a strong predictor of hip fracture7). While our findings are consistent with this previous report7), our study has the advantage of having conducted age-stratified analyses of BMD changes. Contrary to the results of Xu and Wu’s study7) and ours, Cheung et al.8) reported an increase in hip BMD in Hong Kong women aged ≥20 years (mean age, 47 years) across the entire adult age range from 1995-2000 to 2005-2010. Their finding was partly consistent with the long-term reduction in hip fracture incidence in Hong Kong14). In sum, these previous studies showed that long-term changes in adult BMD are associated with changes in hip fracture occurrence. In our study, women in their 40’s and 50’s showed a decreasing BMD trend between 1995 and 2008. It will be informative to observe the changing trend of osteoporotic fracture, including hip fracture, in the next few decades (from 2021) and determine whether the decreasing BMD trend reflects the changes in fracture trend.
Although age-adjusted BMDs of subjects in their 40’s and 50’s decreased during our study period, they did not significantly decrease when further adjusted for BMI. This suggests that the decrease in BMD was due in part to the decrease in BMI. Although the distal forearm, which we used to measure BMD, is not a weight-bearing site, forearm BMD has been reported to correlate well with BMI and body weight15,16).
The secular decrease in BMI is not limited to the present population, but rather is a phenomenon observed throughout Japan. According to the National Health and Nutrition Survey in Japan, the BMI of middle-aged women apparently decreased during our study period17). The long-term decrease in BMI was associated with a combination of decreased energy intake and worsened exercise habits, as well as breakfast skipping17). Important factors associated with low BMI in women include a desire for thinness and dieting behavior18), which are prevalent not only among young women but also among middle-aged and older women19). Moreover, dieting behavior itself has been suggested to have an unfavorable effect on bone strength, including BMD19). The National Health and Nutrition Survey also reported a long-term decrease in calcium intake in middle-aged women, which may be involved in the decrease in BMD we observed17). Thus, controlling dietary factors is a good strategy for bone health maintenance.
We did not observe a significant long-term decrease in BMD of subjects in their 60’s, although their BMIs decreased significantly. While the reason for this inconsistency is unclear, we speculate that menopausal status and healthy lifestyles may have played a role. Women in their 60’s are mostly post-menopausal and experience estrogen insufficiency. However, the extent of decrease in estrogen in post-menopausal women varies, which may have impacted their long-term BMD changes.
Notably, there were some differences between the present female study population and the general Japanese female population with respect to BMI. Mean BMIs reported by the National Health and Nutrition Survey, Japan, 2004 (corresponding to the midpoint of the present study period) were 21.0 (SD 3.0, N=121), 22.6 (SD 3.6, N=127), 23.0 (SD 3.2, N=167), and 23.4 kg/m2 (SD 3.5, N=170) for women in their 30’s, 40’s, 50’s, and 60’s, respectively20). In comparison, BMIs in the present study for women in their 40’s (22.7 kg/m2 and 60’s (22.9 kg/m2) were slightly, but significantly, lower than the national average. These differences may have resulted from the present study population being a health-check population (i.e., those with potentially healthier lifestyles21)). Thus, caution should be exercised when interpreting the results of the present study.
This study has some limitations. First, the decreasing trend in BMD may have been due to selection bias because the number of participants decreased over time during the study period. It is also possible that more women who were worried about their low BMD may have participated in the health check-up examinations later in the study period. Nonetheless, even if there was selection bias, it might not be associated with decreased BMD because the decline in number of subjects after 2008 did not bring about a corresponding decrease in BMD, but rather an increase in BMD. Second, we did not assess secular changes in some age groups given the limited sample size. In particular, changes in BMD of subjects in their 30’s tended to decrease, but not significantly. The 20’s and 30’s are important ages in terms of bone acquisition and maintenance, and thus should be investigated further. Fourth, we could not assess factors other than BMI, such as lifestyles and anti-osteoporotic medications. Nevertheless, their confounding effects may be limited, as evidenced by Japanese community studies22,23) reporting that lifestyle factors are much less influential on adult BMD than BMI (or body weight). With regard to anti-osteoporotic medications that affect BMD, such as bisphosphonates, we previously reported in a community study of BMD22) that 11 of 461 (2.4%) women aged between 60 and 75 years used such medications, whereas none of the women in their 50’s (213 women) used them. Therefore, our results may be biased in terms of the use of anti-osteoporotic medications in women aged over 60 years. Fifth, we conducted several trend tests, which may have resulted in Type I error (i.e., false positives). Finally, we measured cortical BMD, but not spongy BMD. Therefore, while our results apply to osteoporosis of long bones, they may not apply to vertebral bones.
Our study has several implications. First, BMD is an important predictor of osteoporotic fracture, and a good indicator that reflects lifestyle factors. However, long-term trends of BMD have rarely been reported worldwide, and no reports on this topic from Japan have been published. Second, we observed a long-term decrease in BMD in middle-aged women during the period spanning 1995-2013, which could serve as an alert for a potential increase in osteoporosis risk several decades later because hip fracture typically occurs in older adults aged ≥80 years. Finally, although we observed a long-term decrease in BMD being mediated by BMI, the present study design did not allow us to determine a causal association. Nevertheless, such an association has been suggested by previous Japanese cohort studies16,24), and the results of the present study underscore the importance of BMI as a bone mass indicator.