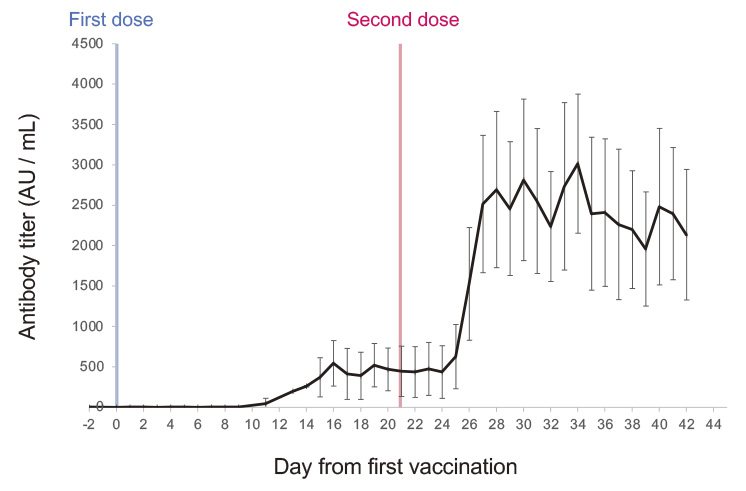
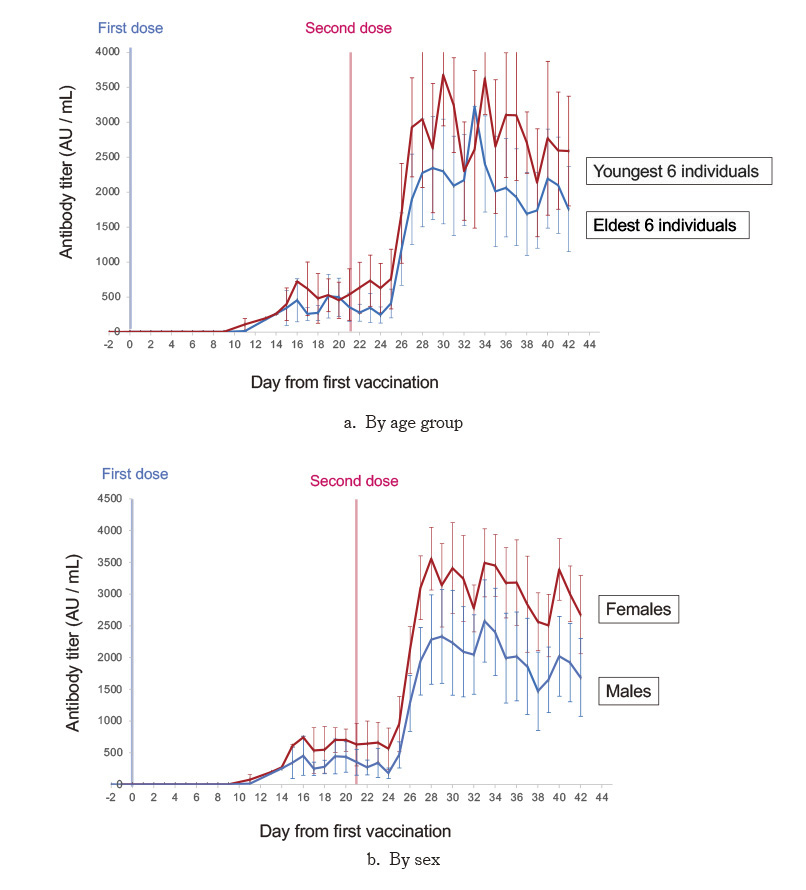
This study investigated the immune response and outcome of BNT162b2 vaccination among 12 staff at a hospital in Fukushima, Japan. Blood samples were collected from participants before their first vaccination, with subsequent sampling performed during the participants’ work days for six weeks thereafter. Antibody titers peaked 6-13 days after the second vaccination (days 27-34 after the first), followed by a steady decrease. Six males had significantly lower peak antibody titers than six females (p= 0.016 witht-test); the older six (median age 53 years) had lower antibody titers than the younger six (median age 35 years) but without statistical significance (pvalue=0.24 witht-test).
Abstract/References
Peak IgG antibody titers against SARS-CoV-2 spike protein following immunization with the Pfizer/BioNTech BNT162b2 vaccine
Yurie Kobashi, Yuzo Shimazu, Takeshi Kawamura, Yoshitaka Nishikawa, Fumiya Omata, Yudai Kaneko, Tatsuhiko Kodama, Masaharu Tsubokura
-
Yurie Kobashi
Department of General Internal Medicine, Hirata Central Hospital, Hirata, Ishikawa District
Department of Radiation Health Management, Fukushima Medical University School of Medicine -
Yuzo Shimazu
Department of Radiation Health Management, Fukushima Medical University School of Medicine
Southern Tohoku Research Institute for Neuroscience -
Takeshi Kawamura
Proteomics Laboratory, Isotope Science Center, The University of Tokyo
Laboratory for Systems Biology and Medicine, Research Center for Advanced Science and Technology, The University of Tokyo -
Yoshitaka Nishikawa
Department of General Internal Medicine, Hirata Central Hospital, Hirata, Ishikawa District
-
Fumiya Omata
Department of General Internal Medicine, Hirata Central Hospital, Hirata, Ishikawa District
-
Yudai Kaneko
Laboratory for Systems Biology and Medicine, Research Center for Advanced Science and Technology, The University of Tokyo
Medical & Biological Laboratories Co., Ltd.
-
Tatsuhiko Kodama
Laboratory for Systems Biology and Medicine, Research Center for Advanced Science and Technology, The University of Tokyo
-
Masaharu Tsubokura
Department of General Internal Medicine, Hirata Central Hospital, Hirata, Ishikawa District
Department of Radiation Health Management, Fukushima Medical University School of Medicine
Abstract
References
1 Hall VJ, Foulkes S, Saei A, et al. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infection (SIREN):a prospective, multicentre, cohort study. Lancet, 397(10286):1725-1735, 2021.
2 Angel Y, Spitzer A, Henig O, et al. Association between vaccination With BNT162b2 and incidence of symptomatic and asymptomatic SARS-CoV-2 infections among health care workers. JAMA, 325(24):2457-2465, 2021.
3 Boyarsky BJ, Werbel WA, Avery RK, et al. Antibody response to 2-dose SARS-CoV-2 mRNA vaccine series in solid organ transplant recipients. JAMA, 325(21):2204-2206, 2021.
Figures (2)